(82-STU) Myeloid-Derived Suppressor Cells Express Diverse Biomarkers Across Tumor Type and are Potential Targets for Cancer-Specific and Personalized Immunotherapies
Friday, October 10, 2025

Mehar F. Nasir, OMS III
Medical Student
Edward Via College of Osteopathic Medicine
Blacksburg, Virginia, United States
Presenter(s)
Introduction/Background: Cancers evade immune detection by creating immunosuppressive tumor microenvironments (TMEs). Tumor cells secrete cytokines and growth factors that drive myeloid precursors to differentiate into myeloid-derived suppressor cells (MDSCs), an immunosuppressive population. In cancer patients, MDSCs accumulate in both TMEs and circulation, where elevated levels are linked to poor prognosis, accelerated disease progression, and decreased survival. MDSCs are divided into monocytic (M-MDSC) and granulocytic (G-MDSC) subtypes based on surface marker expression. Under normal physiology, MDSCs localize to immune-privileged sites such as the placenta and healing wounds, where they suppress T cells and natural killer cells. MDSCs also accumulate in cancer and chronic inflammatory conditions like sepsis and persistent infections. Whether specific tumor types consistently favor certain MDSC subtypes remains unclear. Our research aims to characterize tumors based on the MDSC subtypes present in their microenvironment and to identify whether certain cancers induce MDSC differentiation into M-MDSC, G-MDSC, or both. Our objective is to define tumor-specific MDSC profiles that could serve as biomarkers for immune evasion and guide targeted immunomodulatory therapies.
Methods: We developed a ‘tumor education’ model, an ex vivo tumor-primary CD14+ human monocyte co-culture, to mirror MDSC generation during cancer progression in melanoma and renal cell carcinoma (RCC). We isolated myeloid cells from the co-cultures and performed flow cytometry using antibodies to CD33, CD11b, CD14, CD15, and HLA-DR to evaluate MDSC phenotypes across tumor type and donors.
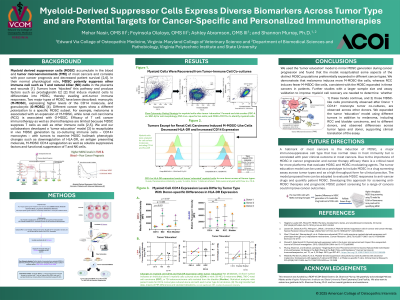
Results/Discussion: We demonstrate that melanoma induces more M-MSDCs, whereas RCC induces comparatively fewer M-MSDCs. These findings are consistent with the observed MDSC profiles in patients with these cancer types.
Conclusions: Our model offers insight into MDSC heterogeneity in cancer and serves as a prototype for screening MDSC profiles in individual patients to inform personalized MDSC-reducing immunotherapies. We are currently expanding the assay to include additional cancer types and a larger donor cohort.
Methods: We developed a ‘tumor education’ model, an ex vivo tumor-primary CD14+ human monocyte co-culture, to mirror MDSC generation during cancer progression in melanoma and renal cell carcinoma (RCC). We isolated myeloid cells from the co-cultures and performed flow cytometry using antibodies to CD33, CD11b, CD14, CD15, and HLA-DR to evaluate MDSC phenotypes across tumor type and donors.
Results/Discussion: We demonstrate that melanoma induces more M-MSDCs, whereas RCC induces comparatively fewer M-MSDCs. These findings are consistent with the observed MDSC profiles in patients with these cancer types.
Conclusions: Our model offers insight into MDSC heterogeneity in cancer and serves as a prototype for screening MDSC profiles in individual patients to inform personalized MDSC-reducing immunotherapies. We are currently expanding the assay to include additional cancer types and a larger donor cohort.
